
A widely-used bridal product has been withdrawn from sale owing to a "serious" chemical hazard. The Office for Product Safety and Standards (OPSS) has issued a product recall notice for the item.
Hania Bridal Henna Paste has been removed from the market by Angel Fashion. The OPSS said: "The product presents a serious chemical risk due to the presence of phenol." Phenol is a prohibited ingredient in cosmetic products. The affected item is described as a green foil 'cone' of henna used in temporary tattooing.
The OPSS said: "Phenol is prohibited in cosmetic products and can cause skin burns and eye damage if applied to the skin.
"Phenol is also toxic if swallowed. Improvements are also required to product labelling and information. The product does not meet the requirements of Regulation (EC) 1223/2009."
Phenol is also known as carbolic acid and hydroxybenzene. It is found in small quantities in cosmetics, paints, polishes, adhesives, lacquers, varnishes, and solvents, reports the Mirror.
The chemical is permitted in consumer products up to 2.5% and soaps and shampoos up to 1% in the EU. It can be used in chemical skin-peelers, nerve injections, topical anaesthetics and as a disinfectant.
However, exposure to high concentrations of phenol can trigger a range of health effects, including irritation, burns and discolouration.
The UK Government says: "Phenol is absorbed into the body very quickly following contact. Effects which may follow include nausea, vomiting, diarrhoea, a fast heart rate and sweating.

"In severe cases drowsiness, breathing and heart problems, lung and kidney damage, and death can occur."
Following the discovery, Angel Fashion has pulled the henna paste from sale.
This comes shortly after a popular beauty cream was recalled from customers due to inaccuracies in ingredient listings and failure to specify fragrance allergens.
Glow and Lovely Hydraglow Rose Enrich Serum Cream was recalled and anyone possessing the product has been urged to cease using it "immediately".
The product is described as a skin cream packaged in a pink-and-white cardboard box and weighing 50g. It carries the barcode 8961014255669.
The OPSS stated: "The product presents a serious chemical risk as the ingredients list had incorrect ingredients listed and had not specified fragrance allergens.
"Consumers who are sensitive to certain allergens are not informed and may experience an adverse effect. The product does not meet the requirements of Regulation (EC) 1223/2009 ('The Cosmetics Regulation')."
It further added: "The product has been withdrawn from the market by Lajawab Foods Limited. We recommend that owners stop using this product immediately and contact the distributor to request redress."
-
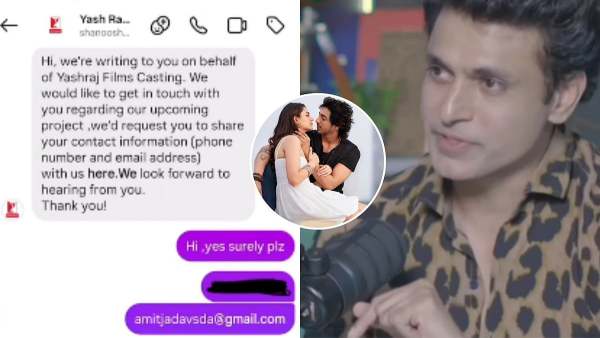
Amit Jadhav Claims Saiyaara Is A Copy Of His 2019 Short Film Khwabon, Seeks Credit: 'Received A Message From YRF Casting In 2022'
-
US–Iran–Israel War: Jaishankar Holds 4th Call With Iran FM Araghchi, Discusses BRICS, Regional Security Amid Strait Of Hormuz Concerns

-
Allu Arjun Slammed For Excluding Raj Kapoor, Ram Gopal Varma & Other Iconic Directors On Allu Cinemas' 'Wall Of Greats'

-
Allu Arjun Slammed For Excluding Raj Kapoor, Ram Gopal Varma & Other Iconic Directors On Allu Cinemas' 'Wall Of Greats'

-
Bengaluru: ED files charge sheet against 19 in TDR scam
